- Understanding electronic and steric effects of ancillary ligands on the catalytic properties of organometallic complexes.
- Synthesis of Ultrafiltration and Nanofiltration Membranes.



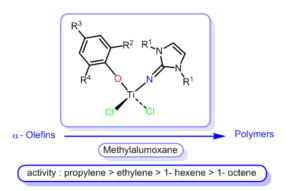 One of our main research topics deal with the synthesis of advanced polymers by novel octahedral homogeneous catalysts This research has focuses on the synthesis, characterization, and the catalytic activity studies for a unique type of coordinative unsaturated group (IV) (Ti, Zr, Hf), “cationic” complexes containing electrophilic ancillary ligands. The types of ancillary ligands, which were pursued for the 1preparation of these cationic complexes, were those containing substituted heteroallylic, acetylacetonato,dikitiminateand imidazoline-2-iminato ancillary ligands (Fig 1). In all cases the ligand motif is bulky enough to prevent solvent coordination. The research is directed in three major simultaneous pathways: (a) The understanding of the electronic effects of the different ancillary ligands on the metal reactivity. (b) The tailoring of chiral and achiral ancillary ligands and chiral or racemic mixtures of complexes, to design a specific stereoregular polymerization of –olefins and design novel chemical transformations to obtain new advanced type of materials. (c) The thoroughly characterization of the new polymeric materials to allow the design of new catalysts for improved polymer properties. The achievement obtained in the synthesis and the application of these new types of coordinative unsaturated complexes are a subject of International basic scientific and technological significance for the novel, clever and unique preparation of new materials.
One of our main research topics deal with the synthesis of advanced polymers by novel octahedral homogeneous catalysts This research has focuses on the synthesis, characterization, and the catalytic activity studies for a unique type of coordinative unsaturated group (IV) (Ti, Zr, Hf), “cationic” complexes containing electrophilic ancillary ligands. The types of ancillary ligands, which were pursued for the 1preparation of these cationic complexes, were those containing substituted heteroallylic, acetylacetonato,dikitiminateand imidazoline-2-iminato ancillary ligands (Fig 1). In all cases the ligand motif is bulky enough to prevent solvent coordination. The research is directed in three major simultaneous pathways: (a) The understanding of the electronic effects of the different ancillary ligands on the metal reactivity. (b) The tailoring of chiral and achiral ancillary ligands and chiral or racemic mixtures of complexes, to design a specific stereoregular polymerization of –olefins and design novel chemical transformations to obtain new advanced type of materials. (c) The thoroughly characterization of the new polymeric materials to allow the design of new catalysts for improved polymer properties. The achievement obtained in the synthesis and the application of these new types of coordinative unsaturated complexes are a subject of International basic scientific and technological significance for the novel, clever and unique preparation of new materials.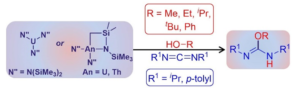 The organometallic chemistry of organoactinides is reaching a high level of sophistication in regards to the basic understanding on the mechanisms by which these complexes operate and have been corroborated with bond disruption enthalpies studies for diverse types of complexes. Our research with organoactinide complexes target their use as catalytic key intermediates in designed catalytic processes with large academic interest. Thus, oriented towards the use of organoimido complexes of actinides to catalyze the anti-Markovnikov addition of an amine to an alkyne to directly synthesized imines. In addition, we have use this complexes for the linear oligomerization of terminal alkynes or the hydrosilylation of terminal alkynes. During the last years, our group has developed new catalytic techniques for the use of actinides in energy demanding catalytic transformations containing oxygen moieties, a myth that was believed to be impossible until few years ago (Fig.2). The unprecedented addition of alcohols to carbodiimides 2mediated by actinide complexes was successfully realized. This represents a rare example of thorium-catalyzed transformations of an alcoholic substrate, and the first example of uranium complexes showing catalytic reactivity with alcohols. Using the uranium and thorium-amides, alcohol additions to unsaturated carbon-nitrogen bonds are achieved in short reaction times with excellent selectivities and high to excellent yields. Computational studies, supported by experimental thermodynamic data, reveal the modes of bonding in the profile of the reaction, which allow the system to overcome the high barrier of scission of the actinide-oxygen bond.
The organometallic chemistry of organoactinides is reaching a high level of sophistication in regards to the basic understanding on the mechanisms by which these complexes operate and have been corroborated with bond disruption enthalpies studies for diverse types of complexes. Our research with organoactinide complexes target their use as catalytic key intermediates in designed catalytic processes with large academic interest. Thus, oriented towards the use of organoimido complexes of actinides to catalyze the anti-Markovnikov addition of an amine to an alkyne to directly synthesized imines. In addition, we have use this complexes for the linear oligomerization of terminal alkynes or the hydrosilylation of terminal alkynes. During the last years, our group has developed new catalytic techniques for the use of actinides in energy demanding catalytic transformations containing oxygen moieties, a myth that was believed to be impossible until few years ago (Fig.2). The unprecedented addition of alcohols to carbodiimides 2mediated by actinide complexes was successfully realized. This represents a rare example of thorium-catalyzed transformations of an alcoholic substrate, and the first example of uranium complexes showing catalytic reactivity with alcohols. Using the uranium and thorium-amides, alcohol additions to unsaturated carbon-nitrogen bonds are achieved in short reaction times with excellent selectivities and high to excellent yields. Computational studies, supported by experimental thermodynamic data, reveal the modes of bonding in the profile of the reaction, which allow the system to overcome the high barrier of scission of the actinide-oxygen bond.1986-1990 – Ph.D. Department of Organic Chemistry, The Hebrew University of Jerusalem, Israel. (Mentor Prof. Jochanan Blum)
1990-1992 – Post – doc, Department of Chemistry, Northwestern University, Evanston IL, U. S. A. (Mentor Prof. Tobin J. Marks)
1993- 1997 Lecturer at the Faculty of Chemistry, Technion Haifa, Israel.
1997- 1999 Senior Lecturer at the Faculty of Chemistry, Technion Haifa, Israel.
1999 – 2003 Associate Professor at the Faculty of Chemistry, Technion Haifa, Israel.
2003- Present Professor at the Faculty of Chemistry, Technion Haifa, Israel.
2006- Present Incumbent of the Samuel O. Friedlander Academic Chair
2008-2012 Dean of the Schulich Faculty of Chemistry
2013- Dean of Student at the Technion
Prizes and Awards
Alexander Goldberg Excellence in Research Award. (1998)
Herschl Rich Technion Innovation Award (1999)
Henri Gutwirth Fund for the Promotion of Research (2000)
L. Meitner, A. v. Humboldt Prize for Research (2001)
Mitchel Soref Research Award (2002)
Henry Taub Research Prize Award (2003)
ECUST China Research Award (2009)
Alexander v. Humboldt Research Prize (2012)
Uzi and Michal Halevy Innovative Applied Engineering Award (2013-2014)
Excellence Teaching Technion Yannai Prize (2013)
| Name | Role | Building/Room | Phone | |
|---|---|---|---|---|
| Dr. Kseniya Kulbitski | ksenia@technion.ac.il | Researchers | 529 | 04-829-3793 |
| Dr. Sayantani Saha | sayantani@tecnion.ac.il | Postdocs | 529 | 04-829-3793 |
| Dr. Hemanta Deka | hemantadghy@gmail.com | Postdocs | 529 | 04-829-3793 |
| Maxim Khononov | khonmax@campus.technion.ac.il | Graduate Students | 529 | 04-829-3793 |
| Alexandra Liobine | sashaka@campus.technion.ac.il | Graduate Students | 529 | 04-829-3793 |
| Konstantin Makarov | makarov@campus.technion.ac.il | Graduate Students | 529 | 04-829-3793 |
| Inbal Ozeri | inbaloz@campus.technion.ac.il | Graduate Students | 529 | 04-829-3793 |
| 419 | 04-829-5033 | |||
| Muhammad Awad | muaawad@campus.technion.ac.il | Graduate Students | 529 | 04-829-3793 |
| Shira Hazan | shira.hazan@campus.technion.ac.il | Graduate Students | 529 | 04-829-3793 |
| Ruibin Wang | ruibin.wang@campus.technion.ac.il | Graduate Students | 529 | 04-829-3793 |
| Cai Liling | ling.cai@campus.technion.ac.il | Graduate Students | 529 | 04-829-3793 |
| Xuhui Zheng | xuhui.zheng@campus.technion.ac.il | Graduate Students | 529 | 04-829-3793 |
| Guy Yardeni | yardeniguy@technion.ac.il | Visiting lecturer | 529 | 04-829-3793 |
| Ebaston Thankarajan-Mini-Mablet | ebastont@campus.technion.ac.il | PostDoc | 529 | 04-829-3793 |
| Sabari Panchavarnam | Sabaritech99@technion.ac.il | 529 | 04-829-3793 | |
| Yue Wang | wang.yue@campus.technion.ac.il | 529 | 04-829-3793 |