Mark Gandelman

CV
Research interests of our group encompass areas of organic, inorganic and organometallic chemistry. We design and develop organometallic and metal-free systems with fundamentally novel properties which can be used as catalysts for important yet problematic chemical reactions. We make a main focus on the understanding of chemical and physical properties of these systems and the mechanistic features of their reactivity. Application of these novel systems to find a solution to challenging catalytic transformations, including asymmetric catalysis, with potential practical importance is our ultimate goal. Ideally, we aim for sustainable, environmentally benign one-pot reactions which generate minimal chemical waste, use non-toxic reagents and require little energy.
Mark Gandelman is Professor of chemistry at the Technion-Israel Institute of Technology, and a vice dean of the teaching affairs. Mark was born and raised in Moldova, and at the age of 18 he immigrated to Israel. He received a B.Sc. degree in chemistry in 1995 from Tel Aviv University, and a Ph.D. degree in 2003 from the Weizmann Institute of Science under the supervision of Prof. David Milstein. Holding the Rothschild fellowship, Mark performed his post-doctoral studies at Harvard University in the group of Prof. Eric N. Jacobsen. In 2005, he joined the Schulich Faculty of Chemistry in the Technion as an assistant professor, and became an associate professor in 2012.
- Nitrenium ions as ligands for transition metals; . Tulchinsky, M. A. Iron, M. Botoshansky, M. Gandelman; Nature Chem. 2011, 3, 525
- Efficient synthesis of secondary alkyl fluorides via Suzuki cross-coupling reaction of 1‑halo‑1‑fluoroalkanes; X. Jiang, S. Sakthivel, K. Kulbitski, G. Nisnevich, M. Gandelman; J. Am. Chem. Soc. 2014, 136, 9548.
- Enantioselective Suzuki cross-couplings of unactivated 1-fluoro-1-haloalkanes: Synthesis of chiral β-, γ-, δ-, and ε-fluoroalkanes. X. Jiang, M. Gandelman. J. Am. Chem. Soc. 2015, 137, 2542.
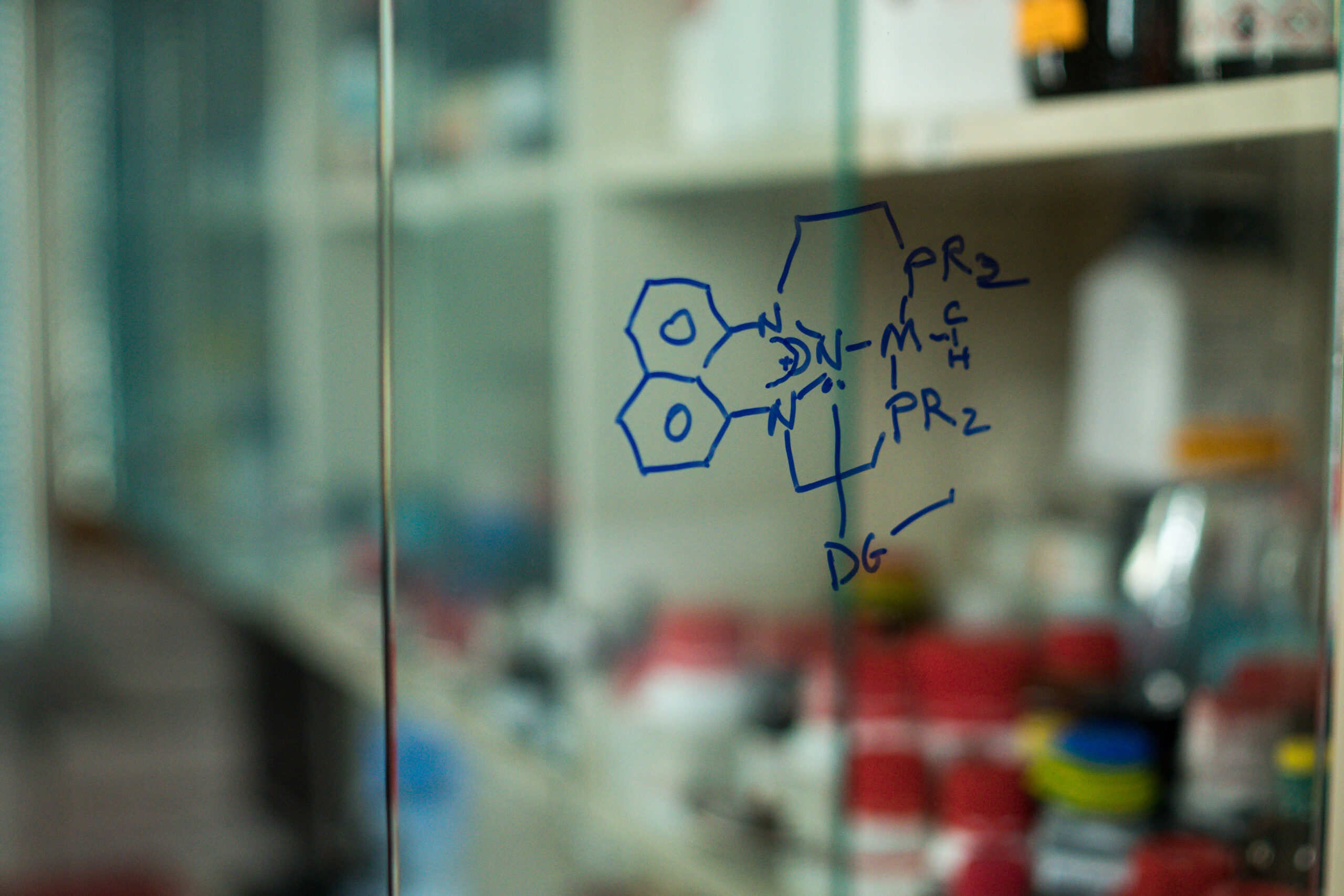
- Nitrogen Lewis Acids. A. Pogoreltsev, Y. Tulchinsky, N. Fridman, M. Gandelman. J.Am. Chem. Soc. 2017, 139, 4062.
- Synthesis of chiral α-trifluoromethyl alcohols and ethers via enantioselective Hiyama cross-couplings of bisfunctionalized electrophiles. A. Varenikov, M. Gandelman. Nature Commun. 2018, 9, 3566.
- Organotitanium nucleophiles in asymmetric cross-coupling reaction: stereoconvergent synthesis of chiral α-fluoroalkyl thioethers. A. Varenikov, M. Gandelman . J.Am. Chem. Soc. 2019, 141, 10994.
- Frustrated Lewis Pairs Comprising Nitrogen Lewis Acids for Si‐H Bond Activation. I. Avigdori, A. Pogoreltsev, A. Kaushanski, N. Fridman, M. Gandelman. Angew. Chem. Int. Ed. 2020, 59, 23476.
- Multimodal Reactivity of N–H Bonds in Triazanes and Isolation of a Triazinyl Radical. A. Koronatov, A. Mauda, B. Tumansky, A. Kaushansky, N. Fridman, D. Bravo-Zhivotovskii, and Gandelman. J. Am. Chem. Soc. 2022, 144, 51, 23642.
| Name | Room | Phone | |
|---|---|---|---|
| Alexander Artaryan | artaryan@technion.ac.il | 528 | 5956 |
| Shoval Meidan Shani | shovalmo@technion.ac.il | 555 | 5951 |
| Alla Pogoreltsev | alka@technion.ac.il | 620 | 3766/3719 |
| Heroot Levy | herootl@technion.ac.il | 528 | 5956 |
| Victoria Bloch | vickis@technion.ac.il | 528 | 5956 |
| Andrii Varenikov | andvar@technion.ac.il | 528 | 5956 |
| Asaf Hassin | sbul@technion.ac.il | 528 | 5956 |
| Idan Avigdori | sidan89@campus.technion.ac.il | 528 | 5956 |
| Liav Leiboviz | liavl@campus.technion.ac.il | 528 | 5956 |
| Moran Levi | smoran22@campus.technion.ac.il | 528 | 5956 |
| Kuldeep Singh | kuldeep@campus.technion.ac.il | 528 | 5956 |
| Deepak Deepak | deepak@campus.technion.ac.il | 528 | 5956 |
| Aleksandr Koronatov | aleksandrk@campus.technion.ac.il | 528 | 5956 |
| Oleg Szumski | oleg.szumski@campus.technion.ac.il | 528 | 5956 |
| Pavel Sakharov | 555 | 5956 |